Fotos / Sonidos
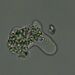
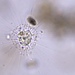
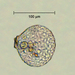
Observ.
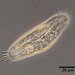
sally3142Descripción
(40x objective) in culture from barky damp soil/puddle sample collected 3 April, 2024
Fotos / Sonidos
Qué
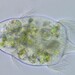
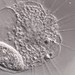
Orden SessilidaObserv.
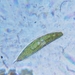
sally3142Descripción
(40x objective) in culture from barky damp soil/puddle sample collected 3 April, 2024
Fotos / Sonidos
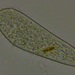
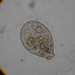
Observ.
sally3142Descripción
(40x objective) in culture from barky damp soil/puddle sample collected 3 April, 2024
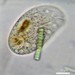
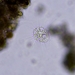
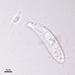
Observ.
tomas_cedhagenDescripción
Distance between crossing lines = 5 mm.
They were picked on the shore.
Fotos / Sonidos
Observ.
peptolabDescripción
Stentor pyriformis Johnson 1893 from the superficial benthos of the river edge of the freshwater segment of estuarine river Peconic River. Imaged in Nomarski DIC on Olympus BH2S using SPlan 10 NA 0.30, SPlanapo 20 NA 0.70, SPlanapo 40 NA 0.95 and SPlan 100 NA1.25 oil objectives plus variable phone camera cropping on Samsung Galaxy S9+. The cells were visible with the naked eye accumulating on the surface of the water at the side of the sample container. Cells measure from 650-800 um when fully extended. The cytoplasm is densely filled with green algal zoochlorella symbionts. There are also much smaller colorless cortical and cytoplasmic granules. The dense population of zoochorellae obscured the internal morphology of the stentors. Macronuclei could only be observed in markedly squashed cells by evaporation of water from under the coverglass. Most cells had two spherical macronuclei, occasionally three. The morphology of my population was identical to that described by Hoshina et al 2021, however their population was smaller than mine at 220-500 um and their macronuclear count also differed from my population which showed 2 MA: "The average number of macronuclei was 6.1 (range 4–10, n = 9) for freshly obtained samples, whereas four-year cultured cells contained only one or two" (1).
"S. pyriformis is a poorly described species, although S. pyriformis is clearly distinguishable from other Stentor species. The species was first described in 1893 and then appeared in a microbiota report in 1908. However, its next appearance was not until 1994, in the study on revision of the genus by Foissner and Wolfl. As described in the original literature, difficulties in the cultivation of this species may have hindered the research on this species. In Japan, S. pyriformis can be found only in highland highly oligotrophic moors, suggesting that intracellular symbiotic algae would help this species of Stentor survive in such a harsh environment. S. pyriformis was described by Johnson in 18936. This algae-bearing Stentor has separated spherical macronuclei without pigmentation, which certainly differentiates it from other Stentor species. While the most common algae-bearing Stentor, S. polymorphus assumes a slender trumpet shape (often shortened), S. pyriformis never resembles such a slender trumpet, but assumes a pear or short conical shape, even when it is swimming. Among algae-bearing Stentor spp., S. polymorphus and S. pyriformis only are considered colorless species, whereas colored species are S. amethystinus, S. fuliginosus, S. araucanus, and S. tartari. Therefore, S. pyriformis is a clearly discernible species; however, it remains underexplored" (1).
"Cells of S. pyriformis were broadly trumpet-shaped, usually 220–500 × 120–300 µm. This length–width ratio did not change significantly between the cells attached to something and swimming. The cells were colored green due to their endosymbiotic green algae that were distributed along the whole body. A large number of tiny transparent vesicles were present along the ciliary rows immediately under the cell surface. To see the contents, the crushed cells were observed. Symbiotic algae appeared to be typical Chlorella-like algae, but no dividing alga was observed. The algal cells appeared more vividly green when compared to those in P. bursaria, suggesting that they are richer in photosynthetic pigments. The symbiotic algae in S. pyriformis had the same size and morphology as those in P. bursaria. Macronuclei were, in general, large and spherical (ø 20–35 µm). The average number of macronuclei was 6.1 (range 4–10, n = 9) for freshly obtained samples, whereas four-year cultured cells contained only one or two. Micronuclei could not be identified" (1).
My population had two macronuclei rarely three in contrast to the populations of Hoshina et al which had an average of 6.1 macronuclei (range 4-10). Also, Hoshina et al 2021 (1) did not identify micronuclei in their multimodality study of S. pyriformis which included electron microscopy. However, the study of Walker 1908 (2) describes the presence of two macronuclei with multiple small micronuclei scattered within the macronucleus. I detected similar small nodules within the macronuclei to those depicted by Walker 1908 but I am uncertain if these are nucleoli or micronuclei. Hishina et al 2021 describe " a large number of transparent vesicles were present along the ciliary rows immediately under the cell surface (1) which I also found. In addition, Hoshina et al 2021 describe numerous starch granules in the cytoplasm which I believe are also present in my samples. These granules are more than twice the size of the subcortical vesicles.
- Characterization of a green Stentor with symbiotic algae growing in an extremely oligotrophic environment and storing large amounts of starch granules in its cytoplasm. Ryo Hoshina, Yuuji Tsukii, Terue Harumoto & Toshinobu Suzaki. Scientific Reports | (2021) 11:2865 | https://doi.org/10.1038/s41598-021-82416-9
- Observations on the Micro-Fauna of an Oregon Pond. Elda R. Walker. Transactions of the American Microscopical Society, Vol. 28, Twenty-Ninth Annual Meeting (Sep., 1908), pp. 75-84
Fotos / Sonidos
Observ.
peptolabDescripción
Cryptomonas (Chilomonas) paramecium Ehrenberg 1832. A bloom from the acidic freshwater kettle pond Chatfield's Hole fed with boiled wheat seeds. Imaged in Nomarski DIC on Olympus BH2S using SPlan 100 1.25 oil objective plus variable phone cropping on Samsung Galaxy S9+. As Jara Kubin points out, the correct name after molecular analyses is Cryptomonas paramecium, the species is a regular member of the genus, but without chlorophyll.
Hoef-Emden, K. & Melkonian, M. (2003). Revision of the genus Cryptomonas (Cryptophyceae): a combination of molecular phylogeny and morphology provides insights into a long-hidden dimorphism. Protist 154: 371-409
Genus: Similar to Cryptomonas in general body form and structure, but colorless; without pyrenoid; "cytopharynx" (gullet) deep, lower half surrounded by granules (ejectisome/trichocyst); one contractile vacuole anterior (Kudo, 1966). Chilomonas does not have chromatophores (pigment-containing structures) and lives by ingesting organic matter.
Species: 20-40 μm long. Posteriorly narrowed; slightly bent 'dorsally'; 30-40 μm long, 10-15 μm wide; widely distributed in stagnant water (Kudo, 1966).
Free-swimming, biflagellate monads, generally obovoid often with a recurved posterior sometimes produced to acutely pointed; not known to produce palmelloid colonies. A longitudinal furrow extends posteriorly from the vestibulum and transforms into a sack-like gullet lined with many rows of ejectosomes. With a single nucleomorph but lacking a chloroplast and pyrenoid; with a prolific accumulation of peripheral starch granules. Periplast with a laminate inner component and a superficial layer of fine fibrillar material. Sexual reproduction is unknown; reproduction being through simple cell division. Cyst formation is unknown.
Fotos / Sonidos
Qué
Stentor igneusObserv.
peptolabDescripción
Stentor igneus Ehrenberg, 1838 from the acidic freshwater kettle pond Chatfield's Hole after feeding the culture with boiled wheat seeds. Imaged in Nomarski DIC on Olympus BH2S using SPlanapo 40 0.95 and SPlan 100 1.25 oil objectives plus variable phone cropping on Samsung Galaxy S9+.
Rose-colored or colorless; macronucleus oval; ciliation uniform. This small, reddish-pigmented freshwater species is well defined due to the redescriptions by Foissner (1980) and Song and Wilbert (1989). Foissner et al. (1992) reviewed the data available on its morphology and ecology. Stentor igneus lacks symbiotic algae and has two synonyms: S. roseus and S. ruber. Measurements. 200-400 um long.
Fotos / Sonidos
Observ.
juanloredoDescripción
Water sample collected from the river San Pedro, Congregación Ortíz, Rosales, Chihuahua, at the indicated location.
The sample was collected on January 5th at 15:42. The images were acquired on January 7th.
Fotos / Sonidos
Observ.
amayakanDescripción
Video (x5 speed): https://youtu.be/0qTAE73Mgt4
Photos are snapshots of the same individual in the video
Size: see scale bar
Site of collection: Pavilion, Takamori Higashi Park (a freshwater habitat), Sendai, Japan
Date of collection: Aug. 8th 2021
Weather: Rainy
Water temp.: 29.0°C
pH 6.6
Date of observation: September 12th 2021 (the collected sample in a plastic container was left near a window out of direct sunlight at room temperature until observation).
Bright field observation using a Wraymer microscope (model BX-3500TL, Osaka, Japan) equipped with a Floyd-2 HDMI ethernet digital camera (Wraymer, Osaka, Japan). The accuracy of the scale bar was confirmed by using a stage micrometer glass slide (1 div. = 10 µm; Wraymer, Osaka, Japan) at each magnification.
Fotos / Sonidos
Observ.
amayakanDescripción
Video: https://youtu.be/NVS2DjYjfX8
Photos are snapshots of the same individual in the video.
The cell measures 77 µm x 30 µm
Site of collection: Katsurashima Ryokuchi south pond (a freshwater habitat), Sendai, Japan
Date of collection: February 11th 2022
Weather: Sunny
Water temp.: 5.2°C
pH 6.6
Date of observation: February 12th 2022 (the collected sample in a plastic container was left near a window out of direct sunlight at room temperature until observation)
Bright field observation using a Wraymer microscope (model BX-3500TL, Osaka, Japan) equipped with a Floyd-2 HDMI ethernet digital camera (Wraymer, Osaka, Japan).
Fotos / Sonidos
Observ.
amayakanFecha
Septiembre 18, 2021Descripción
video (x1 speed): https://youtu.be/wCv0HsU80Bo
Photos are snapshots of the same individual in the video
Cell size: 50 µm x 43 µm
Site of collection: Katsurashima Ryokuchi south pond (a freshwater habitat), Sendai, Japan
Date of sample collection: September 18th 2021
Weather: Rainy
Water temp.: 21.3°C
pH 6.7
Date of observation: September 23rd 2021 (the collected sample in a plastic container was left near a window out of direct sunlight at room temperature until observation)
Bright field observation using a Wraymer microscope (model BX-3500TL, Osaka, Japan) equipped with a Floyd-2 HDMI ethernet digital camera (Wraymer, Osaka, Japan). The accuracy of the scale bar, which was set according to the guidance of the manual of the software (Spectman) provided for the camera, was confirmed by using a stage micrometer glass slide (1 div. = 10 µm; Wraymer, Osaka, Japan) at each magnification.
Qué
Género NebelaObserv.
nickbelliveauDescripción
From mosses and liverworts in damp roadside ditch. 400x magnification.
Qué
Género NebelaObserv.
nickbelliveauDescripción
From vernal pool on a bed of Sphagnum sp. 400x magnification.
Fotos / Sonidos
Observ.
peptolabDescripción
Acineria uncinata Tucolesco, 1962 from the freshwater pond Kellis Pond which appeared after feeding the culture with boiled wheat seed. Imaged in Nomarski DIC on Olympus BH2 using SPlan 100 1.25 oil objective plus variable phone camera cropping on Samsung Galaxy S9+. The cell measures 48 um in length. There are three visible somatic kineties which differentiates this species from A. incurvata which has 11 somatic kineties. Augustin, Foissner & Adam 1987 describe 3 right kineties while Martin-Cereceda, Serrano and Guinea 1995 describe 4 (rarely 5)- 2 left and 2 right. This, and the presence of the short oral slit being restricted to the anterior pole are discriminatory. There are two spherical macronuclei with a large intercalated micronucleus between them.
"Acineria uncinata is a lanceolate ciliate with a short oral slit restricted to the anterior region, and rolled up forming a characteristic overlapping in the anterior area at the left side of the cell. Sizes range between 26-38 um in length and 11-17 um in width in OsO4 fixed specimens. The nuclear apparatus consists of two nearly spherical macronuclei, closely together, and one large micronucleus between them. A single contractile vacuole is at the posterior end of the cell. Extrusomes are regularly placed at the anterior end of the cell, and other are dispersed at the cytoplasm. Somatic infraciliature is composed of four kineties (rarely five) which present a different arrangement at both sides of the cell. The two right ones (rk1, rk2) have long cilia and show kinetodesmal fibers, while the two left somatic kineties (lk1, lk2; a third left kinety is sometimes present) possibly lack such fibers. A kinetal segment with short clavate cilia, composed of pairs of kinetosomes, is located at the left dorsal side. Three perioral kineties are located at both sides of the oral region. Left perioral kinety (pk1) shows kinetosomes with slight fibers which could correspond to transverse microtubules. At right side, two kineties accompany the oral slit, one of them (pk2), short in length, seems to be formed by pairs of kinetosomes whose fibrillar derivates did not stain, while the other kinety (pk3), extending to the posterior end of the cells, is formed by kinetosomes with long cilia and conspicuous kinetodesmal fibers. This perioral kinety is thus very likely a somatic ciliary row except for its anterior region" (1).
"Acineria uncinata Tucolesco, 1962 Brackish and freshwater. About 35-55 um. Body lanceolate without lateral edge. Anterior pole overlapping towards the left side. Two spherical macronuclei showing a clearer zone at their central region. Sometimes a single, elongated, tapered nucleus. Contractile vacuole terminal, often surrounded by a group of smaller vacuoles. Cytostome a straight and short slit restricted to the rolled up anterior pole. Can therefore feed only on small prey (flagellates). Three somatic kineties on the right side with 20-22 cilia each. Cilia at the ventral margin of the anterior third transformed to regularly curved crotchets. Occurence and Ecology. This species was found in summer 1954 in a small dirty brackish puddle near Lake Tekirghiol and in mesosaprobic freshwaters of Bucharest. Tucolesco (1962) separated this species from A. incurvata by the non-overlapping post-oral dorsal margin. However, in A. incurvata the situation is rather similar. Thus, we propose the following characters for discrimination from A. incurvata: the presence of only three somatic kineties on the right side, the (probably) unciliated left side, and the short oral slit being restricted to the anterior pole. Unmentioned since description. Note after proof reading: This is a valid species which we rediscovered recently" (2).
- Mercedes Martin-Cereceda, Susana Serrano, and Almudena Guinea; Occurrence of Acineria uncinata in Activated Sludge Plants: Morphology and Relationship with Physico-Chemical Plant Parameters. Arch.Protistenkd. 146:79-84, 1995
- Revision of the genera Acineria, Trimyema and Trochiliopsis (Protozoa, Ciliophora). H. Augustin, W. Foissner & H. Adam. Bull.Br.Mus.Nat.Hist. (Zool) 1987 52(6): 197-224
Fotos / Sonidos
Observ.
sally3142Descripción
culture from temporary mud puddles and wet soil with algae in old orchard land W of Tavira, Portugal; collected 3 April 2024
Video on my post to fb group Amateur Microscopy: https://www.facebook.com/1300443920/videos/pcb.2592299670951432/800319331606585
Estadísticas
- 7505